
 Copyright © Michael Richmond.
This work is licensed under a Creative Commons License.
Copyright © Michael Richmond.
This work is licensed under a Creative Commons License.
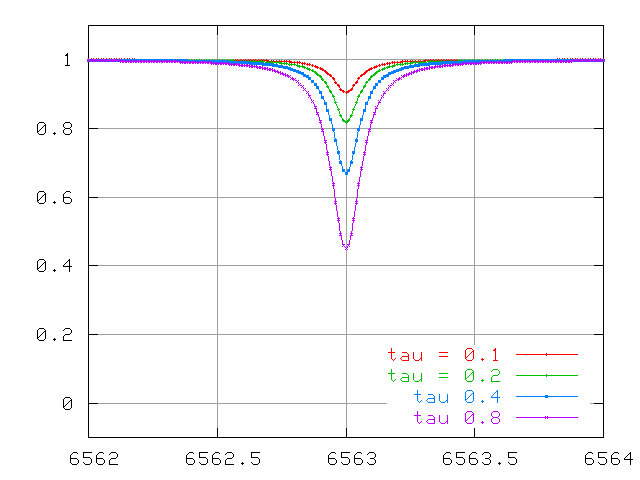
So, we measure a stellar spectrum, and notice a very weak absorption line. This happens to correspond to an absorption line which has an optical depth τ = 0.1. That means that most of the photons of the central wavelength (6563 Angstroms in the example below) produced within the photosphere manage to escape before being scattered or absorbed.

For ordinary stars like the Sun, weak lines are dominated by Doppler broadening. The width of the line is caused by random motions of the atoms absorbing the light. How does Doppler broadening grow as we increase the number of absorbing atoms?
Recall that the optical depth can be defined as
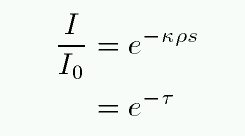
So, if we double the density ρ (and hence number of atoms), we double the optical depth. How does this affect the equivalent width? For thermal broadening, one can calculate the change in the absorption profile and find ....
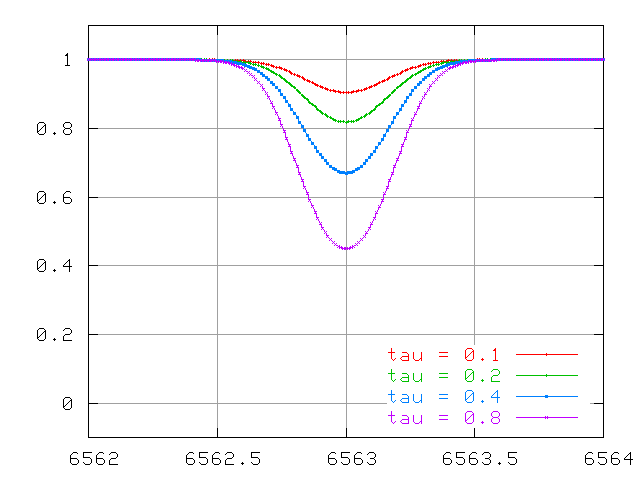
At first, this doubling of the optical depth causes a doubling of the the equivalent width of the line
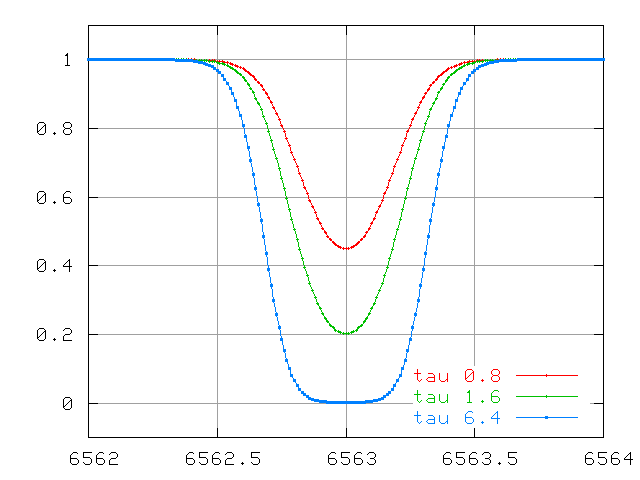
But as the line becomes optically thick, the equivalent width no longer grows in step with the optical depth (and number of atoms):
And for lines which are VERY optically thick, the equivalent width due to thermal motions grows very slowly ....
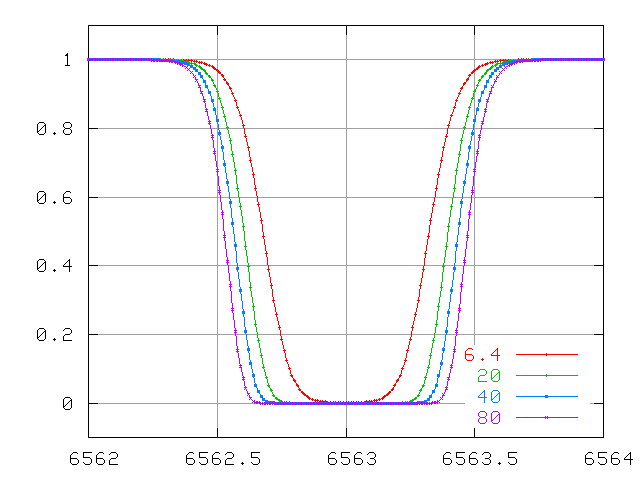
The problem here is that the only way the equivalent width can increase is for the wings to grow. The wings of this line are due to atoms which are moving either towards or away from the observer. How many atoms are moving really fast? The distributions of speeds in an ordinary cloud of gas obey a Maxwellian distribution:
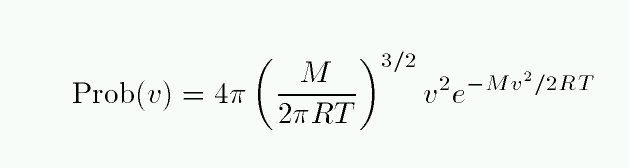
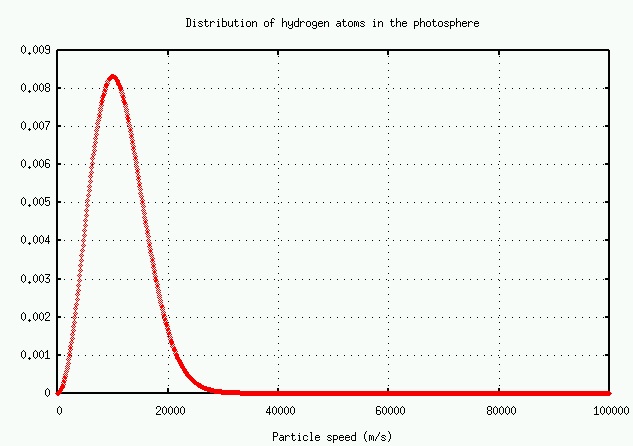
The number of atoms with speed v falls off at high speeds as
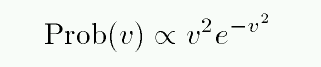
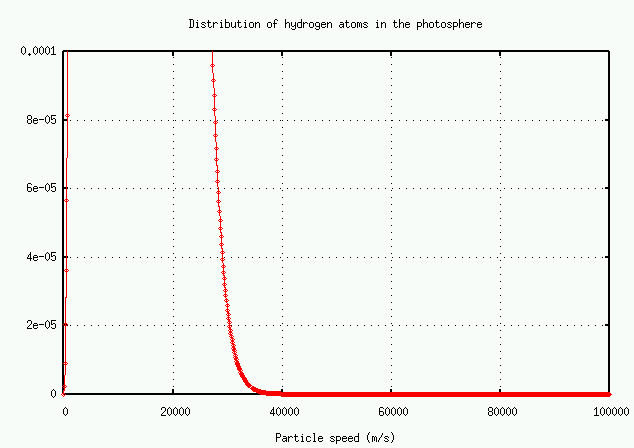
It's easier to read the numbers on a logarithmic plot:
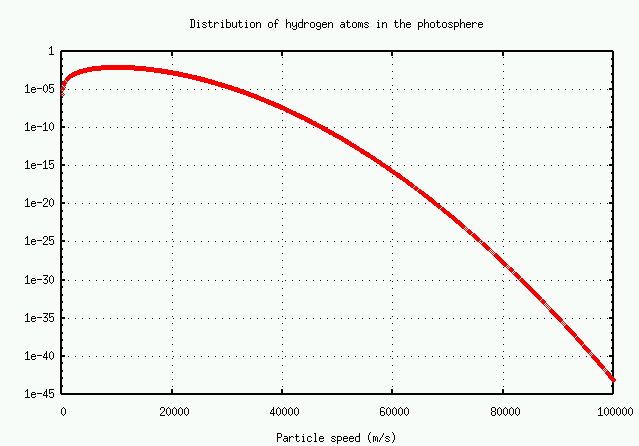
In brief, the number of atoms with very high speeds -- and hence very large Doppler shifts -- decreases very rapidly. There just aren't many atoms available to grow the wings.
Exercise:There are very, very roughly 10^(38) hydrogen atoms in the entire solar photosphere. Using the graph above, estimate
Remember, these are TOTAL number of atoms in the entire photosphere!
- the typical speed of a hydrogen atom
- the total number of hydrogen atoms with this speed
- the Doppler shift in an H-alpha line due to this motion
- the total number of hydrogen atoms which would cause a Doppler shift twice as large
- the total number of hydrogen atoms which would cause a Doppler shift four times as large
- the total number of hydrogen atoms which would cause a Doppler shift eight times as large
The pressure and collisional (and the natural broadening) processes all produce a Lorentzian line profile: the optical depth τ changes with wavelength as
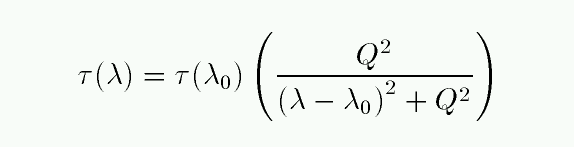
At large distances from the line center, this optical depth goes like

There is no exponential involved this time, so the function falls off much more slowly than that for thermal broadening. As a result, the lines have much wider wings.
Watch the lines change at small optical depth ...
... optical depths of order unity ....
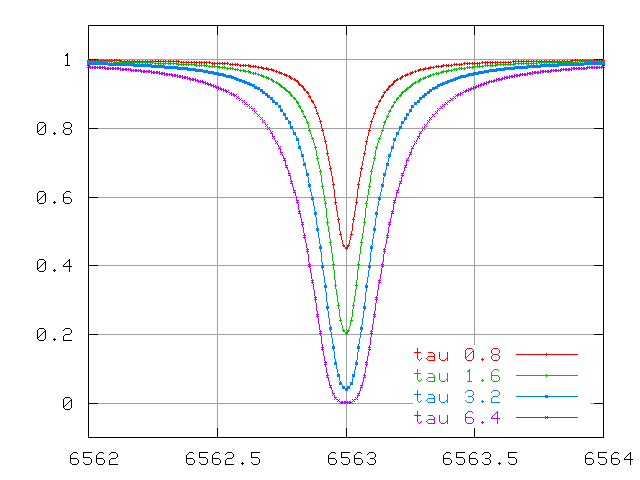
... and very large optical depths.
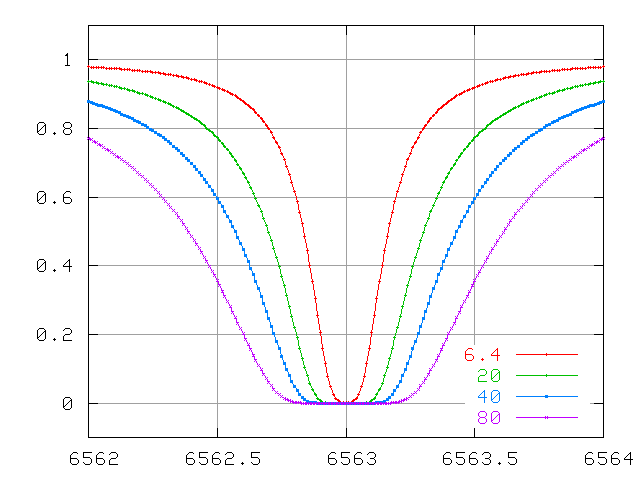
The wings continue to increase in width quickly, even after the line becomes saturated.
Compare the behavior of Doppler-broadened and collisionally-broadened lines at small optical depth
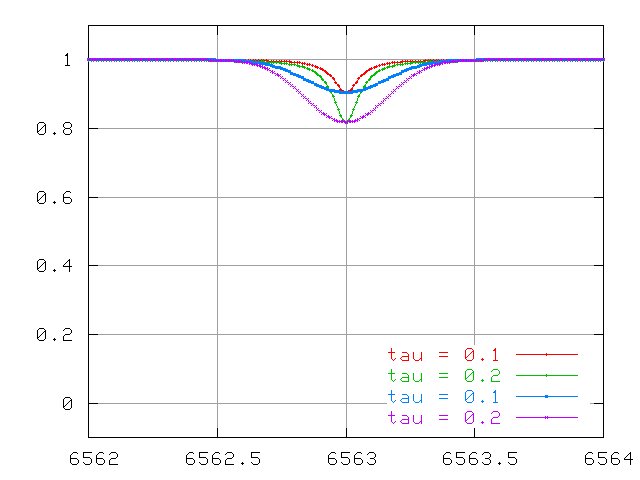
and at large optical depth.

The combination of both Doppler and collisional processes leads to a complex change in the equivalent width of a spectral line as the optical depth increases. Let's use N to denote the number of absorbing atoms, and W the equivalent width of the line.
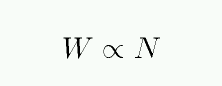
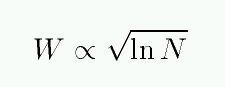
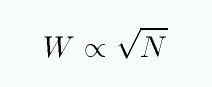
A plot of the equivalent width W as a function of the number of absorbing atoms N is called the curve of growth.

This figure taken from Carroll and Ostlie,
"An Introduction to Modern Astrophysics" (Addison-Wesley 1996)
You can see the three different regimes: optically thin, saturated, and REALLY saturated.
If you look closely at the figure above, you'll see that the axes are a bit peculiar.
So, how do we use this? The basic idea is
Let's try it! Below is a section of the spectrum near the 5890 line of neutral sodium, produced when a sodium atom jumps from the ground state 3s1 to the excited state 3p1.
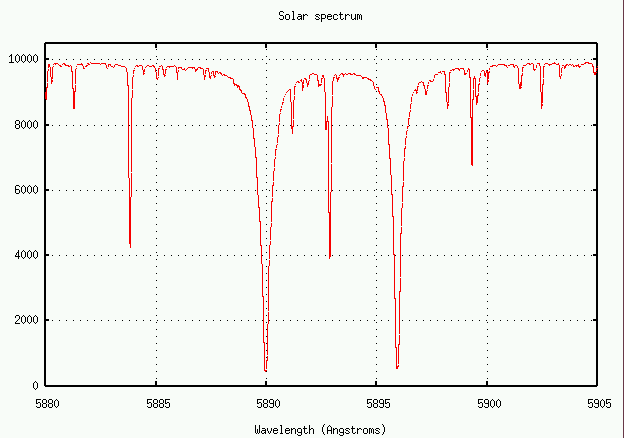
The oscillator strength for this transition is f = 0.645.
Exercise:
- Measure the equivalent width of this sodium absorption line
- Find its location on the curve of growth
- Calculate the column density of sodium atoms in the ground state
- Estimate the ratio of sodium atoms in the ground state to sodium atoms in excited states
- Estimate the ratio of neutral sodium atoms to ionized sodium atoms; you'll need to know the partition functions for neutral and ionized sodium (2.4 and 1.0, respectively), and the ionization energy for sodium (about 5.1 eV).
- Compute the total column density of sodium atoms in the sun's photosphere.
Note: in class, the column density below was misprinted: it was a factor of 1000 too large
- The column density of hydrogen atoms is about 6.6 x 10^(23); what is the abundance of sodium relative to hydrogen?
 Copyright © Michael Richmond.
This work is licensed under a Creative Commons License.
Copyright © Michael Richmond.
This work is licensed under a Creative Commons License.